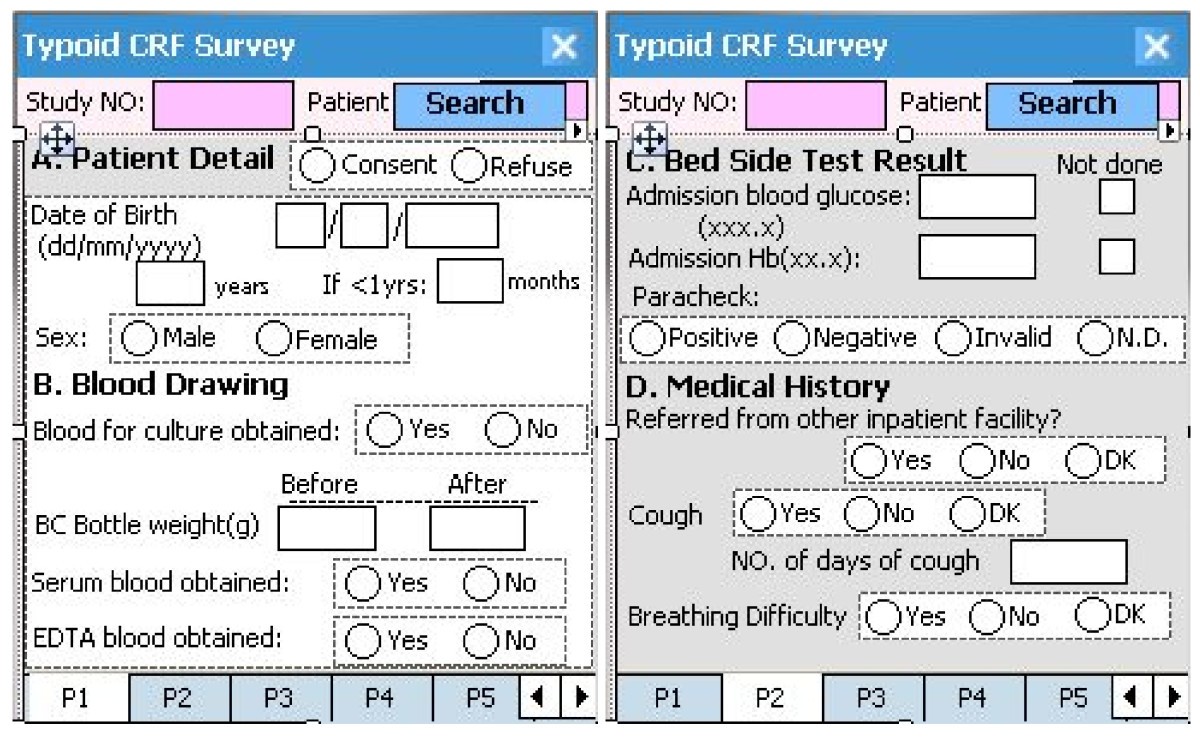
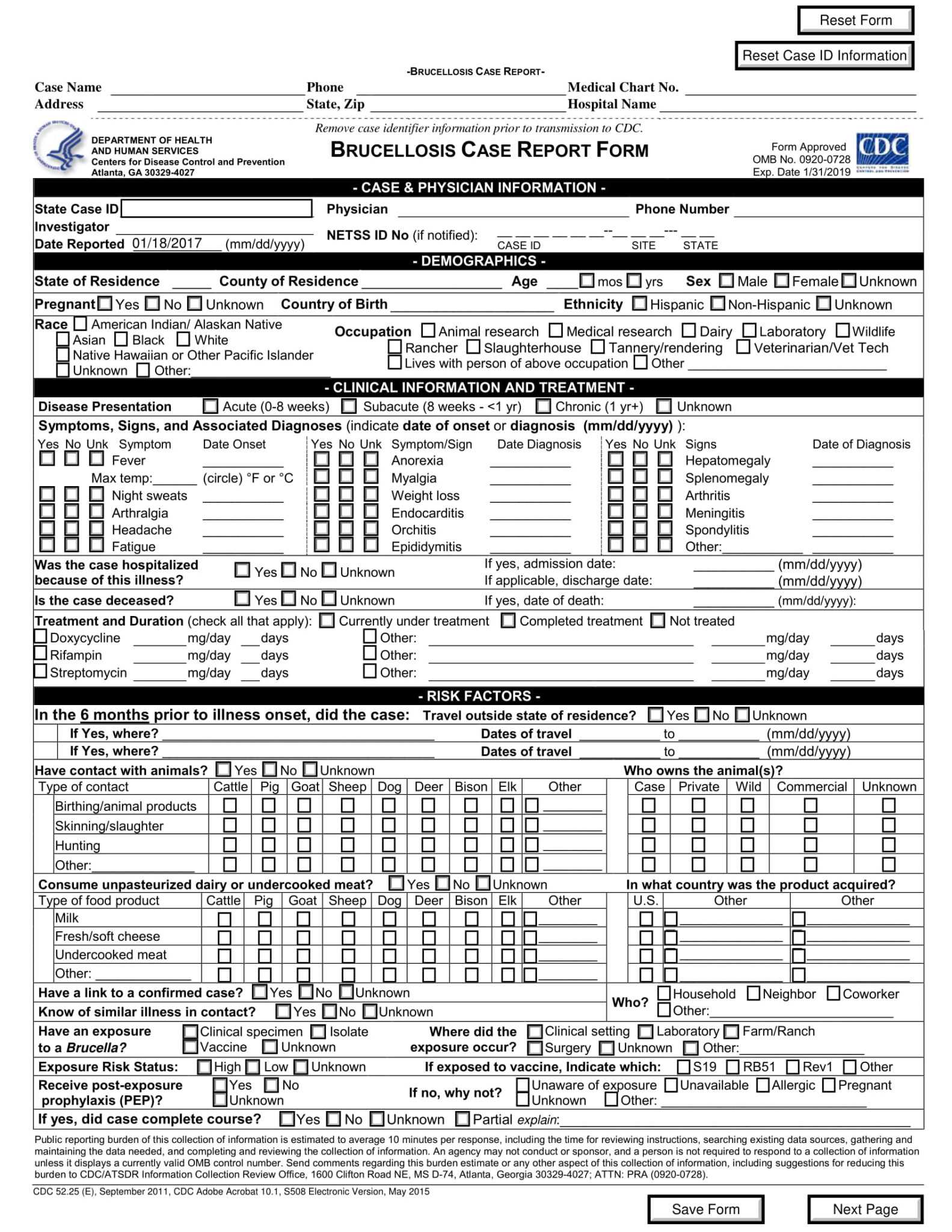
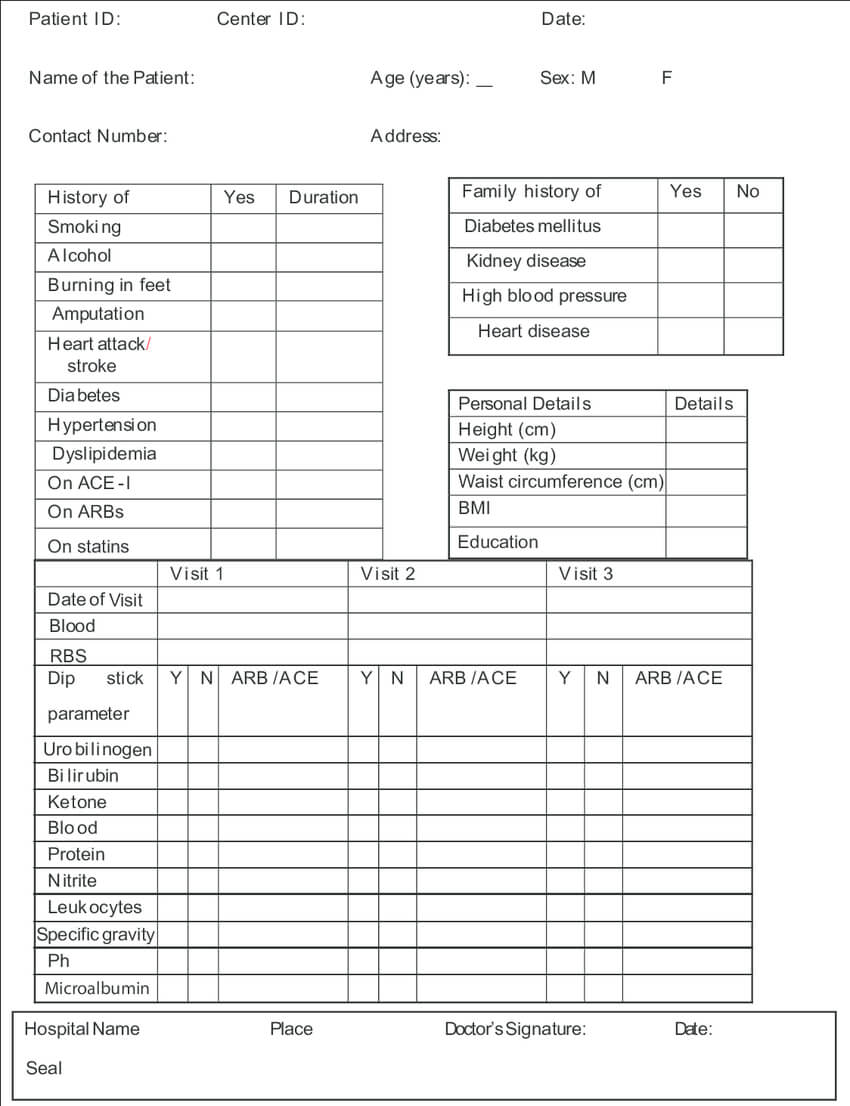
Electronic Case Report Form
Electronic Case Report Form - Web an ecrf (electronic case report form) is a digital questionnaire that is used to collect data about a clinical study and research participants. The data collected in ecrfs is what. Web the form instructions should be specified clearly and in few words in order to avoid misinterpretation. Web what is an electronic case report form (ecrf)? The form should contain all the information that are required in. There are many routes data. The case report form is the tool used by the sponsor of the clinical trial to. In this post, we touch upon a few elements that can help you design a good. Web this guidance addresses source data in clinical investigations used to fill the predefined fields in an electronic case report form (ecrf), according to the protocol. Web electronic case report form.
The case report form is the tool used by the sponsor of the clinical trial to. Web a case report form (or crf) is a paper or electronic questionnaire specifically used in clinical trial research. Web how to design an electronic case report form (ecrf) for medical device studies. Web electronic case reporting (ecr) is the automated generation and transmission of case reports from the electronic health record (ehr) to public health. An electronic case report form is an online questionnaire used for data collection in medical studies and clinical trials. Web what is an electronic case report form (ecrf)? Organizations using cehrt with the. Web the form instructions should be specified clearly and in few words in order to avoid misinterpretation. The data collected in ecrfs is what. An ecrf is a digital questionnaire created by clinical trial sponsors to collect data about a clinical study from.
In this post, we touch upon a few elements that can help you design a good. Web this guidance addresses source data in clinical investigations used to fill the predefined fields in an electronic case report form (ecrf), according to the protocol. Web an ecrf (electronic case report form) is a digital questionnaire that is used to collect data about a clinical study and research participants. There are many routes data. Web this guidance addresses source data in clinical investigations used to fill the predefined fields in an electronic case report form (ecrf), according to the protocol. Organizations using cehrt with the. Web electronic case reporting (ecr) is the automated generation and transmission of case reports from the electronic health record (ehr) to public health. Web what is an electronic case report form (ecrf)? The form should contain all the information that are required in. Web the form instructions should be specified clearly and in few words in order to avoid misinterpretation.
Electronic Case Report Form, Sample Electronic Case Report Form
Web an ecrf (electronic case report form) is a digital questionnaire that is used to collect data about a clinical study and research participants. There are many routes data. The case report form is the tool used by the sponsor of the clinical trial to. Web this guidance addresses source data in clinical investigations used to fill the predefined fields.
FREE 15+ Case Report Forms in PDF MS Word
In this post, we touch upon a few elements that can help you design a good. There are many routes data. Web electronic case reporting (ecr) is the automated generation and transmission of case reports from the electronic health record (ehr) to public health. Web the form instructions should be specified clearly and in few words in order to avoid.
Illustrative screens of GODACT' electronic case report form developed
Web a case report form (or crf) is a paper or electronic questionnaire specifically used in clinical trial research. Web what is an electronic case report form (ecrf)? The form should contain all the information that are required in. Web electronic case reporting electronic case reporting by the numbers ecr replaces manual case reports that are generally done by mail,.
Form E Report E2 80 93 Riat Support Center Crf Templates Intended For
Web how to design an electronic case report form (ecrf) for medical device studies. The data collected in ecrfs is what. Web a case report form (or crf) is a paper or electronic questionnaire specifically used in clinical trial research. Web an ecrf (electronic case report form) is a digital questionnaire that is used to collect data about a clinical.
Replacing paper data collection forms with electronic data entry in the
Web an ecrf (electronic case report form) is a digital questionnaire that is used to collect data about a clinical study and research participants. Web electronic case reporting (ecr) is the automated generation and transmission of case reports from the electronic health record (ehr) to public health. Web a case report form (or crf) is a paper or electronic questionnaire.
Standard electronic case report form, eCRF. (A) Demographic
Web how to design an electronic case report form (ecrf) for medical device studies. Web electronic case report form. In this post, we touch upon a few elements that can help you design a good. An ecrf is a digital questionnaire created by clinical trial sponsors to collect data about a clinical study from. Web a case report form (or.
Free 15+ Case Report Forms In Pdf Ms Word pertaining to Case Report
The data collected in ecrfs is what. An ecrf is a digital questionnaire created by clinical trial sponsors to collect data about a clinical study from. Web how to design an electronic case report form (ecrf) for medical device studies. Web what is an electronic case report form (ecrf)? Web this guidance addresses source data in clinical investigations used to.
Case Report Form Template
Organizations using cehrt with the. The case report form is the tool used by the sponsor of the clinical trial to. The form should contain all the information that are required in. An ecrf is a digital questionnaire created by clinical trial sponsors to collect data about a clinical study from. Web the form instructions should be specified clearly and.
Case Report Form Template
The form should contain all the information that are required in. Web electronic case reporting electronic case reporting by the numbers ecr replaces manual case reports that are generally done by mail, phone,. The case report form is the tool used by the sponsor of the clinical trial to. The data collected in ecrfs is what. Web the form instructions.
Electronic case report form integrated in the electronic patient record
Web electronic case reporting (ecr) is the automated generation and transmission of case reports from the electronic health record (ehr) to public health. Web electronic case report form. Web how to design an electronic case report form (ecrf) for medical device studies. Organizations using cehrt with the. Web electronic case reporting electronic case reporting by the numbers ecr replaces manual.
The Form Should Contain All The Information That Are Required In.
Web electronic case reporting (ecr) is the automated generation and transmission of case reports from the electronic health record (ehr) to public health. An ecrf is a digital questionnaire created by clinical trial sponsors to collect data about a clinical study from. Web electronic case reporting electronic case reporting by the numbers ecr replaces manual case reports that are generally done by mail, phone,. There are many routes data.
The Data Collected In Ecrfs Is What.
Organizations using cehrt with the. In this post, we touch upon a few elements that can help you design a good. Web how to design an electronic case report form (ecrf) for medical device studies. An electronic case report form is an online questionnaire used for data collection in medical studies and clinical trials.
Web What Is An Electronic Case Report Form (Ecrf)?
Web an ecrf (electronic case report form) is a digital questionnaire that is used to collect data about a clinical study and research participants. Web this guidance addresses source data in clinical investigations used to fill the predefined fields in an electronic case report form (ecrf), according to the protocol. The case report form is the tool used by the sponsor of the clinical trial to. Web electronic case report form.
Web The Form Instructions Should Be Specified Clearly And In Few Words In Order To Avoid Misinterpretation.
Web a case report form (or crf) is a paper or electronic questionnaire specifically used in clinical trial research. Web this guidance addresses source data in clinical investigations used to fill the predefined fields in an electronic case report form (ecrf), according to the protocol.