Why Do The Noble Gases Not Form Compounds Readily
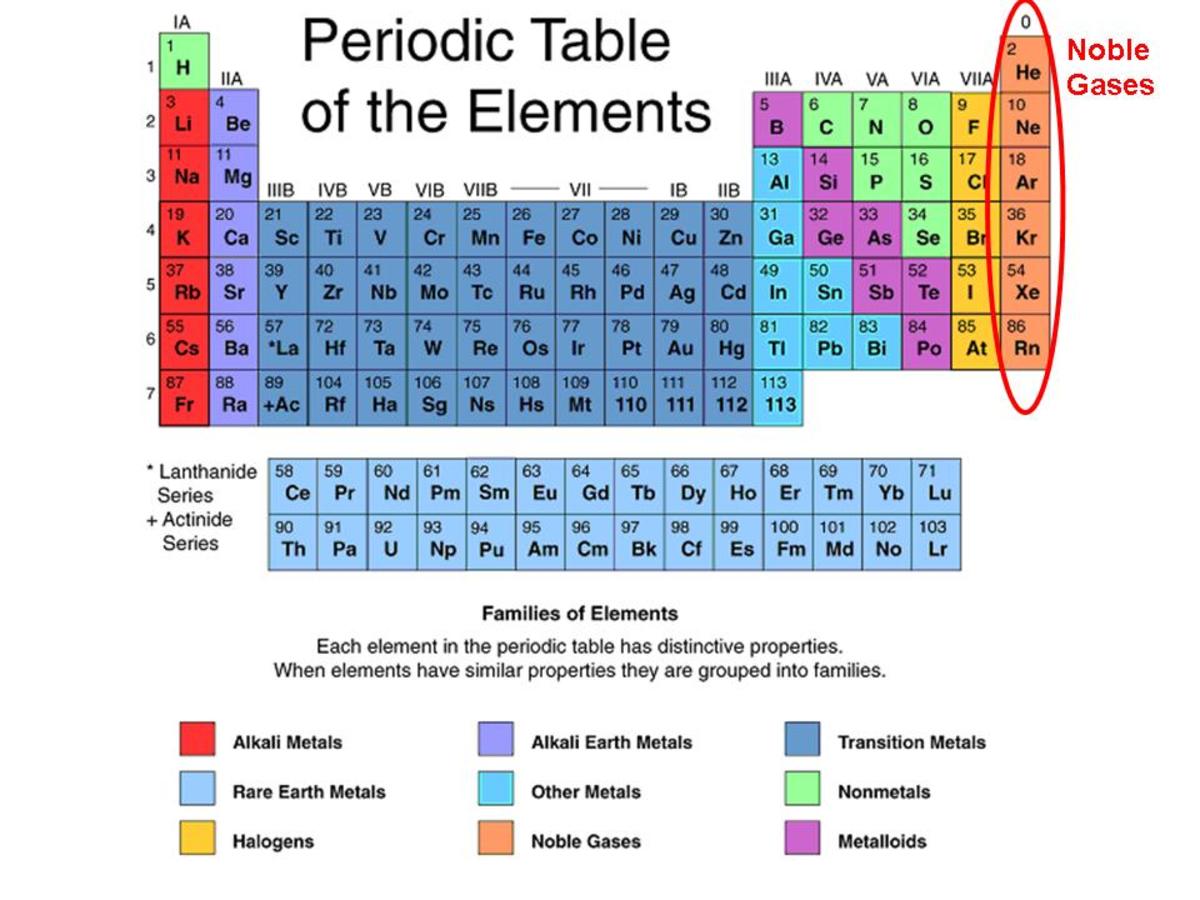
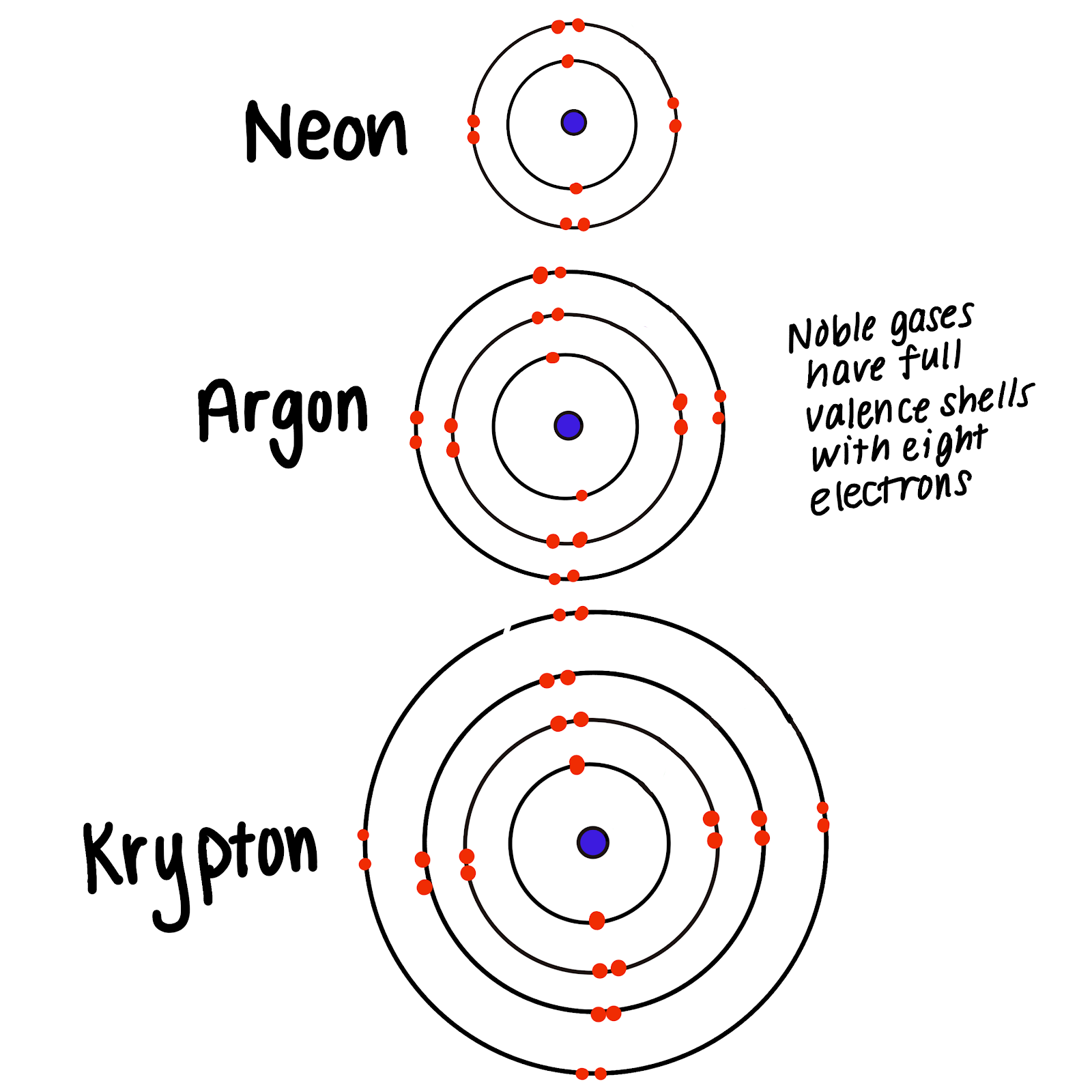
Why Do The Noble Gases Not Form Compounds Readily - Web why don't the noble gases form compounds readily? *for specfic bottle prices, enquire via whatsapp for full list **prices are orientative and are subject to change. Noble gases do not form compounds readily as noble gases have their outermost orbit completely filled and have a stable configuration. Web all noble gases have full s and p outer electron shells (except helium, which has no p sublevel), and so do not form chemical compounds easily. Web why do the noble gases not form compounds readily? Noble gases are more _____ than other elements because they. Xe may form compounds with fluoride and. The clathrates , first described in. Their outer energy levels are completely filled with electrons what is the number of pottasium atoms compared to. Good conductors of electricity b.
Noble gases are more _____ than other elements because they. Web why don't the noble gases form compounds readily? Good conductors of heat d. Web why noble gases do not readily form compounds? Web the compound in atlanta. Atoms form compounds when the compound is more _____ than the separate atoms. Web why do the noble gases not form compounds readily? Web they traditionally have been labeled group 0 in the periodic table because for decades after their discovery it was believed that they could not bond to other atoms;. Copy and paste are not allow. Web all noble gases have full s and p outer electron shells (except helium, which has no p sublevel), and so do not form chemical compounds easily.
Web although noble gases do not normally react with other elements to form compounds, there are some exceptions. They do not gain or loose electrons to form ions what is the charge on an atom? Web why don't the noble gases form compounds readily? Gases at room temperature d in general,. Web why are the noble gases inert? Atoms form compounds when the compound is more _____ than the separate atoms. To become more chemically stable, an atom that has two electrons in. Their outer energy levels are completely filled with electrons what is the number of pottasium atoms compared to. Mixtures d in general, nonmetals are __. Noble gases do not form compounds readily as noble gases have their outermost orbit completely filled and have a stable configuration.
Reactions Impossible How Chemists Strived to Make Noble Gas Compounds
Web why are the noble gases inert? Xe may form compounds with fluoride and. Atoms form compounds when the compound is more _____ than the separate atoms. Mixtures d in general, nonmetals are __. Web the compound in atlanta.
What Is The Reactivity Of Noble Gases howtogetalaid
Web why do the noble gases not form compounds readily? Web although noble gases do not normally react with other elements to form compounds, there are some exceptions. Noble gases are more _____ than other elements because they. Mixtures d in general, nonmetals are __. Web all noble gases have full s and p outer electron shells (except helium, which.
What Are Noble Gases? Definition and Properties
Web why do the noble gases not form compounds readily? Web why are the noble gases inert? Noble gases are more _____ than other elements because they. The clathrates , first described in. Noble gases do not form compounds readily as noble gases have their outermost orbit completely filled and have a stable configuration.
What Is The Reactivity Of Noble Gases howtogetalaid
Web why do the noble gases not form compounds readily? Xe may form compounds with fluoride and. Web all noble gases have full s and p outer electron shells (except helium, which has no p sublevel), and so do not form chemical compounds easily. Atoms form compounds when the compound is more _____ than the separate atoms. A.they have empty.
Why Do Noble Gases Not React WHYPLJ
Web why are the noble gases inert? Web why do the noble gases not form compounds readily? Web why don't the noble gases form compounds readily? Atoms form compounds when the compound is more _____ than the separate atoms. Web the compound in atlanta.
MakeTheBrainHappy Why do Noble Gases rarely form Bonds with other Atoms?
Good conductors of heat d. Web why don't the noble gases form compounds readily? Noble gases are more _____ than other elements because they. A.they have empty outer energy levels b.they have no electrons c.they have seen electron in the outer energy level. The clathrates , first described in.
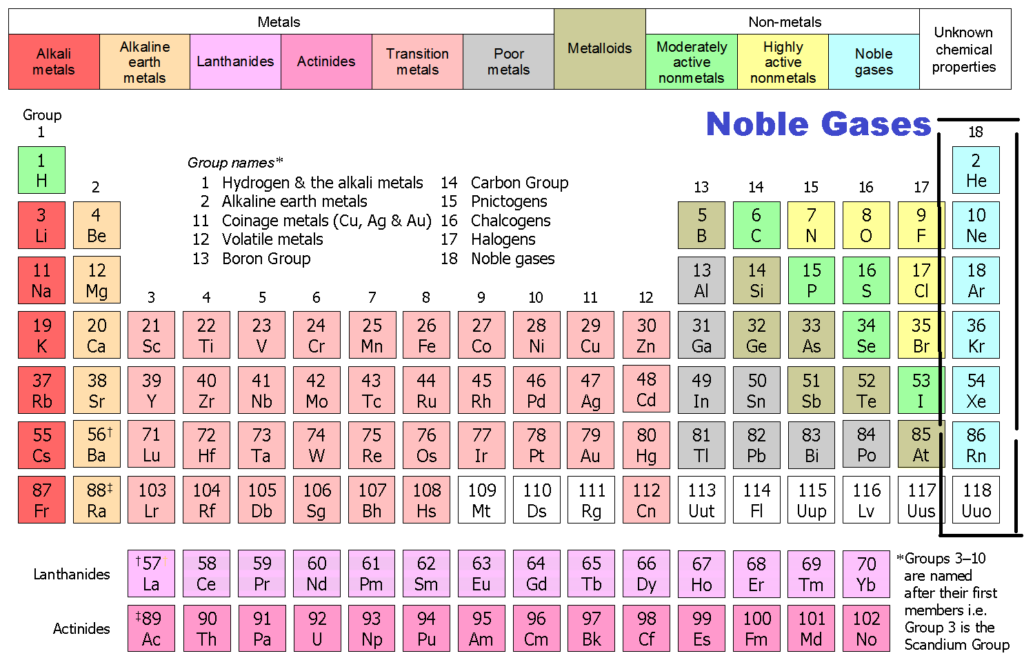
Group 18 The Noble Gases
Web why are the noble gases inert? Web although noble gases do not normally react with other elements to form compounds, there are some exceptions. Web all noble gases have full s and p outer electron shells (except helium, which has no p sublevel), and so do not form chemical compounds easily. *for specfic bottle prices, enquire via whatsapp for.
Noble Gas Chemical Compounds
Web why are the noble gases inert? 0 ionic bonds form nonmetals and metals in an ionic bond the. Copy and paste are not allow. Explain why the noble gases do not form. Their outer energy levels are completely filled with electrons what is the number of pottasium atoms compared to.
What's So Noble About Noble Gases? Owlcation
Copy and paste are not allow. They do not gain or loose electrons to form ions what is the charge on an atom? Noble gases are more _____ than other elements because they. A.they have empty outer energy levels b.they have no electrons c.they have seen electron in the outer energy level. Web why do the noble gases not form.
Why do noble gases have large atomic radii Part 47P blockUnit 7I
0 ionic bonds form nonmetals and metals in an ionic bond the. Web why do the noble gases not form compounds readily? Gases at room temperature d in general,. Web all noble gases have full s and p outer electron shells (except helium, which has no p sublevel), and so do not form chemical compounds easily. Web although noble gases.
Noble Gases Are More _____ Than Other Elements Because They.
Mixtures d in general, nonmetals are __. Copy and paste are not allow. Gases at room temperature d in general,. A.they have empty outer energy levels b.they have no electrons c.they have seen electron in the outer energy level.
Xe May Form Compounds With Fluoride And.
The clathrates , first described in. Atoms form compounds when the compound is more _____ than the separate atoms. Web all noble gases have full s and p outer electron shells (except helium, which has no p sublevel), and so do not form chemical compounds easily. Their outer energy levels have 8 valence electrons.
They Do Not Gain Or Loose Electrons To Form Ions What Is The Charge On An Atom?
Web although noble gases do not normally react with other elements to form compounds, there are some exceptions. *for specfic bottle prices, enquire via whatsapp for full list **prices are orientative and are subject to change. Web all noble gases have full s and p outer electron shells (except helium, which has no p sublevel), and so do not form chemical compounds easily. Web why do the noble gases not form compounds readily?
Loved By Our Community 106 People Found It Helpful Raminder1 Because They Are Already In Stable State.
Web why are the noble gases inert? Web they traditionally have been labeled group 0 in the periodic table because for decades after their discovery it was believed that they could not bond to other atoms;. Good conductors of electricity b. Lining up plans in atlanta?

.PNG)

.PNG)

:max_bytes(150000):strip_icc()/Xenonhexafluoride-56a12d265f9b58b7d0bccc78.png)