How Do Metals Form Ions
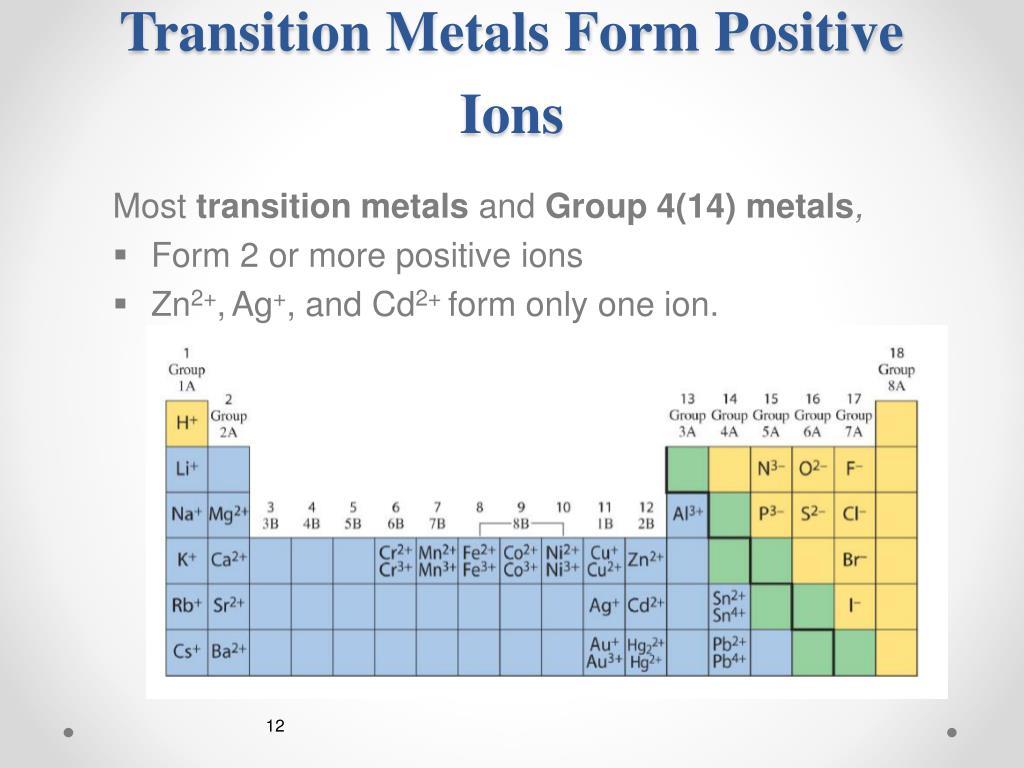
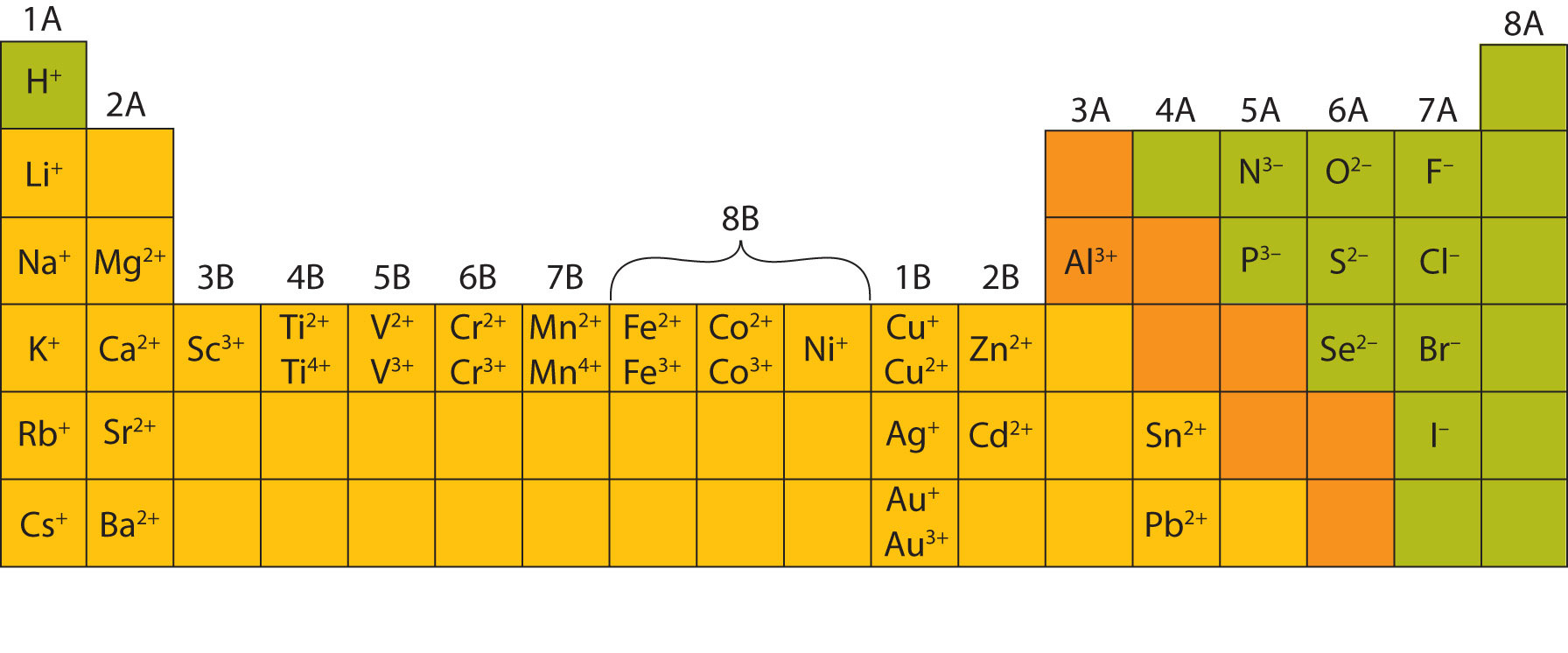
How Do Metals Form Ions - A metal ion in aqueous solution or aqua ion is a cation, dissolved in water, of chemical formula [m (h 2 o) n] z+. Granite is a type of rock that forms from magma. Web transition metals have unfilled inner \(d\) electron shells. Web metal atoms lose electrons from their outer shell when they form ions: Ions form primarily through loss of \(s\) electrons. Web this is actually one of the chemical properties of metals and nonmetals: Many transition metals can form more than one ion. Web you’ll notice under ‘formation of ions’ that the transition metals react to form ions with different charges. A geode is a rounded, hollow rock that is often lined with mineral crystals. Metals tend to form cations, while nonmetals tend to form anions.
Web the minerals in the original rock were formed at one set of conditions, but were then subjected to different conditions of heat, pressure, and h 2 o abundance in earth’s crust. Web in general, when metals form an ion, it is easier for them to lose electrons to attain a stable noble gas electronic configuration, as most metals have electrons. Many transition metals can form more than one ion. Web minerals are found on the earth's crust and mantle and many were created when feldspar and quartz reacted with other materials during the formation of the planet. Web this is actually one of the chemical properties of metals and nonmetals: Web how do minerals form from water solutions? Web most metals become cations when they make ionic compounds. Web ions are formed by the addition of electrons to, or the removal of electrons from, neutral atoms or molecules or other ions; Second, most atoms form ions of a single. Web metal atoms lose electrons from their outer shell when they form ions:
Web how do metals form ions? Web minerals form under an enormous range of geologic conditions. Web in general, when metals form an ion, it is easier for them to lose electrons to attain a stable noble gas electronic configuration, as most metals have electrons. To obtain a full outer shell: Web ions are formed when atoms lose or gain negatively charged electrons. By combination of ions with other. An iron (ii) ion has a 2+ charge, and an iron (iii) ion has a 3+ charge. There are probably more ways to form minerals than there are types of minerals themselves. The number lost is the same as. Web ions are formed by the addition of electrons to, or the removal of electrons from, neutral atoms or molecules or other ions;
Ions Predict Charge Stone Cold Chemistry Talk Ions Predict Charge
Metals tend to form cations, while nonmetals tend to form anions. Web minerals form under an enormous range of geologic conditions. The ions are positive, because they have more protons than electrons the ions formed have full outer shells the. Web minerals are found on the earth's crust and mantle and many were created when feldspar and quartz reacted with.
Chem matters ch6_ionic_bond
Web ions form when atoms lose or gain electrons. Web metal ions in aqueous solution. Web ions are formed when atoms lose or gain negatively charged electrons. Some atoms have nearly eight electrons in their valence shell and can gain additional valence electrons until they. Many transition metals can form more than one ion.
Write My Paper For Me how to write ions
Metals tend to form cations, while nonmetals tend to form anions. An iron (ii) ion has a 2+ charge, and an iron (iii) ion has a 3+ charge. The number lost is the same as. It is formed in the way that all minerals generally are. Web ions form when atoms lose or gain electrons.
PPT Naming Ionic Compounds PowerPoint Presentation, free download
By combination of ions with other. Web the minerals in the original rock were formed at one set of conditions, but were then subjected to different conditions of heat, pressure, and h 2 o abundance in earth’s crust. Web you’ll notice under ‘formation of ions’ that the transition metals react to form ions with different charges. A geode is a.
CH105 Chapter 3 Ionic and Covelent Bonding Chemistry
Web how do minerals form from water solutions? Web this is actually one of the chemical properties of metals and nonmetals: By combination of ions with other. Web how do metals form ions? Web minerals are found on the earth's crust and mantle and many were created when feldspar and quartz reacted with other materials during the formation of the.
Chem matters ch6_ionic_bond
Web metal ions in aqueous solution. Web ions form when atoms lose or gain electrons. An iron (ii) ion has a 2+ charge, and an iron (iii) ion has a 3+ charge. It is formed in the way that all minerals generally are. A metal ion in aqueous solution or aqua ion is a cation, dissolved in water, of chemical.
PPT How do atoms form ions? PowerPoint Presentation ID7021047
Web you’ll notice under ‘formation of ions’ that the transition metals react to form ions with different charges. Some atoms have nearly eight electrons in their valence shell and can gain additional valence electrons until they. A geode is a rounded, hollow rock that is often lined with mineral crystals. Metals tend to form cations, while nonmetals tend to form.
Ions
It contains the minerals quartz (clear), plagioclase feldspar (shiny white), potassium feldspar (pink), and. Web in general, when metals form an ion, it is easier for them to lose electrons to attain a stable noble gas electronic configuration, as most metals have electrons. Web most metals become cations when they make ionic compounds. Web how do metals form ions? The.
5.2.1 Formation of Ion Revision.my
Web ions form when atoms lose or gain electrons. Web minerals form under an enormous range of geologic conditions. Second, most atoms form ions of a single. Web ions are formed by the addition of electrons to, or the removal of electrons from, neutral atoms or molecules or other ions; Web you’ll notice under ‘formation of ions’ that the transition.
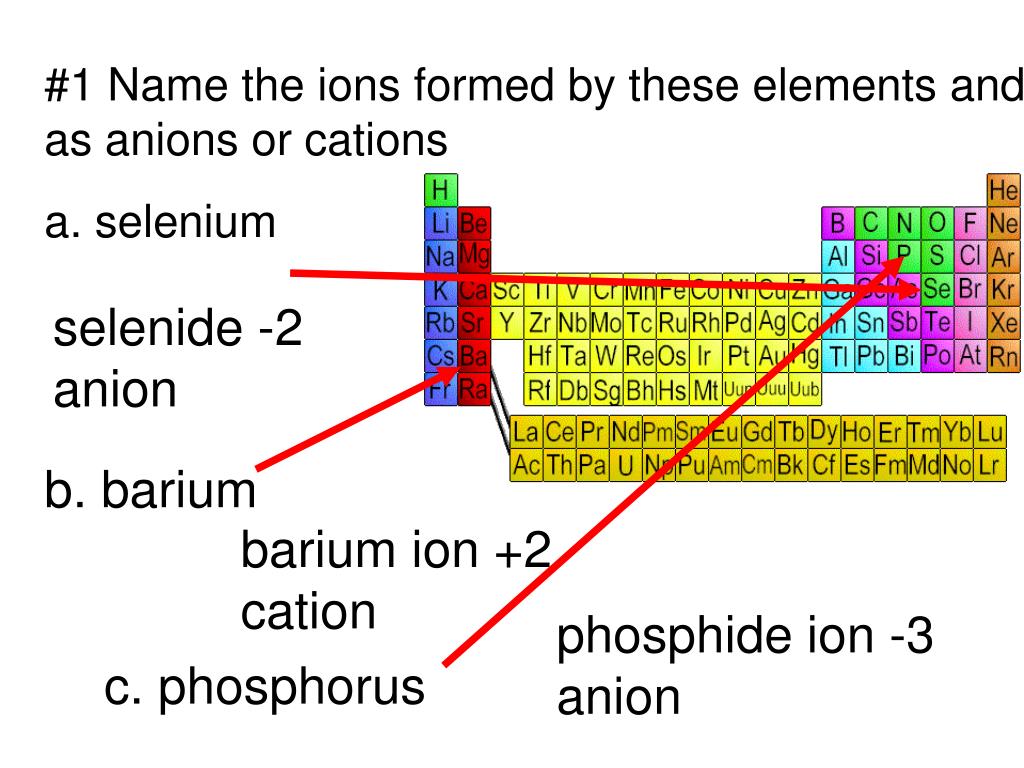
PPT 1 Name the ions formed by these elements and classify them as
Ions form primarily through loss of \(s\) electrons. Metals form electopositive ions or cations. It contains the minerals quartz (clear), plagioclase feldspar (shiny white), potassium feldspar (pink), and. Web most metals become cations when they make ionic compounds. The ions are positive, because they have more protons than electrons the ions formed have full outer shells the.
Metals Tend To Form Cations, While Nonmetals Tend To Form Anions.
Web minerals are found on the earth's crust and mantle and many were created when feldspar and quartz reacted with other materials during the formation of the planet. Web the minerals in the original rock were formed at one set of conditions, but were then subjected to different conditions of heat, pressure, and h 2 o abundance in earth’s crust. Web how do metals form ions? To obtain a full outer shell:
Metals Tend To Form Cations, While Nonmetals Tend To Form Anions.
By combination of ions with other. Web you’ll notice under ‘formation of ions’ that the transition metals react to form ions with different charges. Second, most atoms form ions of a single. Web in general, when metals form an ion, it is easier for them to lose electrons to attain a stable noble gas electronic configuration, as most metals have electrons.
The Number Of Electrons Lost Or Gained Determines The Charge Of The Ion:
Web metal ions in aqueous solution. A geode is a rounded, hollow rock that is often lined with mineral crystals. An iron (ii) ion has a 2+ charge, and an iron (iii) ion has a 3+ charge. It contains the minerals quartz (clear), plagioclase feldspar (shiny white), potassium feldspar (pink), and.
Metals Form Electopositive Ions Or Cations.
Ions form primarily through loss of \(s\) electrons. Web most metals become cations when they make ionic compounds. Web how do minerals form from water solutions? The number lost is the same as.